What we do today
defines what’s possible tomorrow.
We’re not just striving to protect vision – we’re helping shape the future of pediatric care. Sydnexis was founded on a simple but urgent truth: pediatric progressive myopia is a degenerative condition that demands early, targeted intervention.
Myopia is one of the most common eye diseases in children and the numbers are rising at an unprecedented rate. More than a refractive condition, it carries long-term risks for irreversible vision complications. We believe early intervention can change that trajectory and we are honored to be leading that charge. Because the choices we make today don’t just impact vision now – they shape visual future for generations.
What we do today
defines what’s possible tomorrow.
We’re not just striving to protect vision – we’re helping shape the future of pediatric care. Sydnexis was founded on a simple but urgent truth: pediatric progressive myopia is a degenerative condition that demands early, targeted intervention.
Myopia is one of the most common eye diseases in children and the numbers are rising at an unprecedented rate. More than a refractive condition, it carries long-term risks for irreversible vision complications. We believe early intervention can change that trajectory and we are honored to be leading that charge. Because the choices we make today don’t just impact vision now – they shape visual future for generations.
A Landmark Therapy in Development
We are developing SYD-101 to help address one of the most urgent unmet needs in pediatric ophthalmology.
SYD-101 is a proprietary low-dose atropine formulation currently being evaluated in a landmark Phase 3 clinical trial. Engineered specifically for growing eyes, this investigational therapy aims to slow myopia progression and reduce the risk of long-term ocular complications.

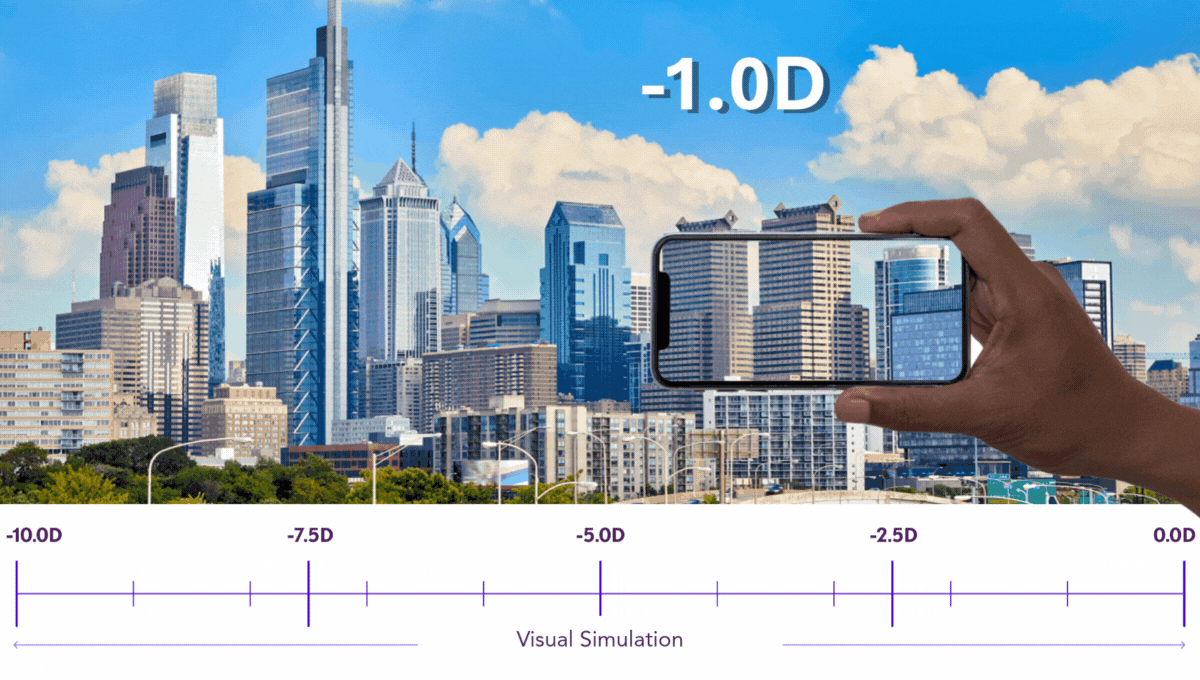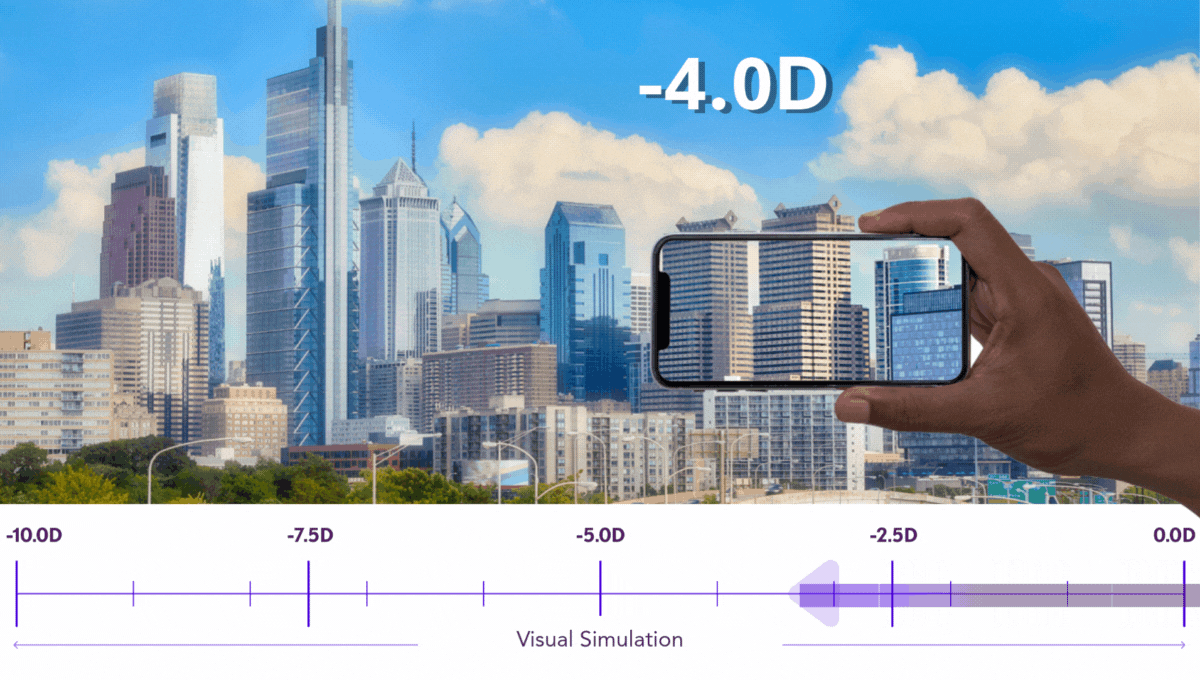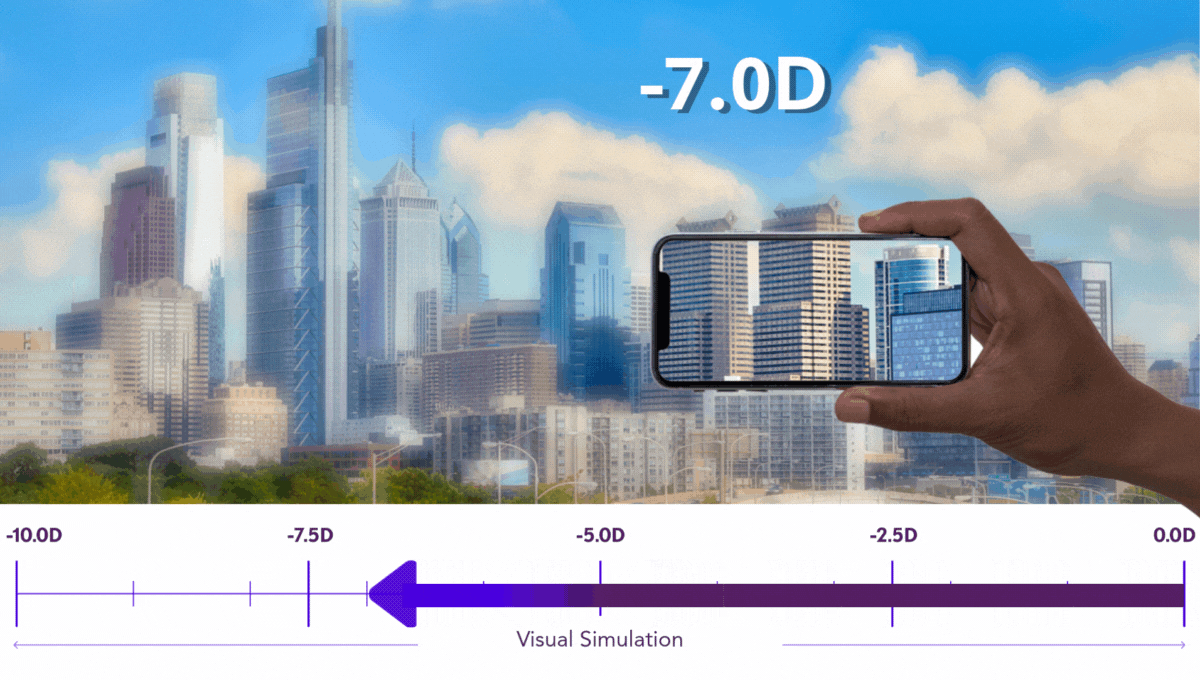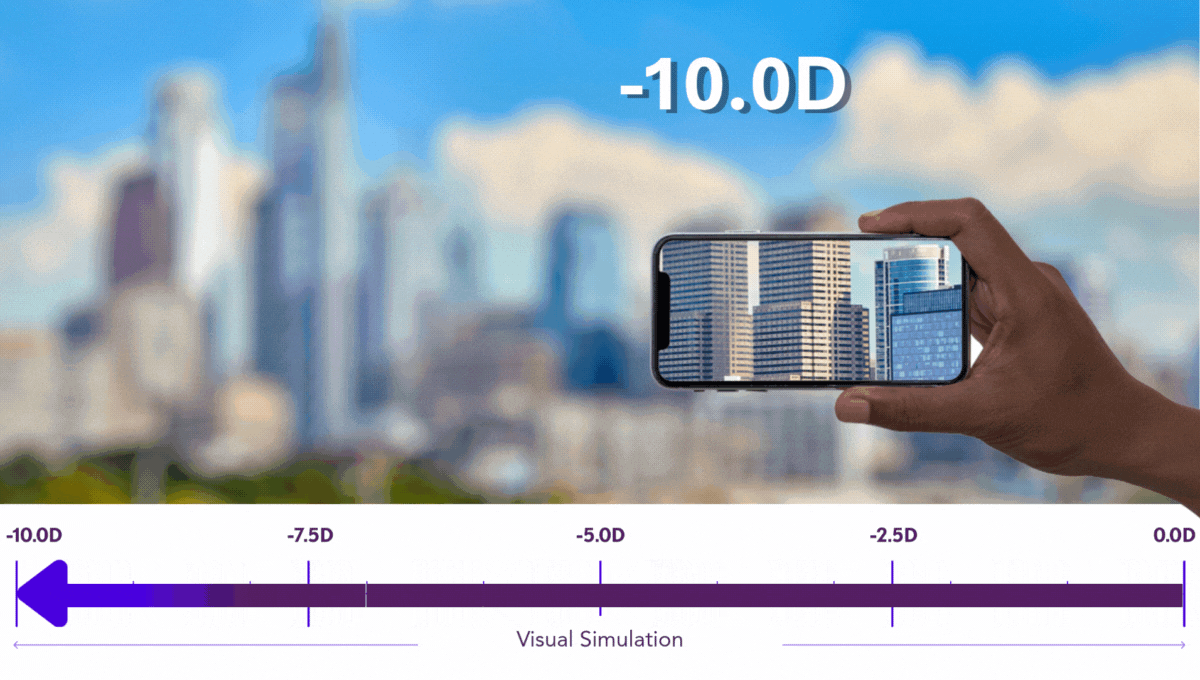
Preserving Vision is Critical for Kids
Pediatric progressive myopia isn’t just a refractive issue. It’s a degenerative condition with lifelong implications.
Our work is dedicated to preserving every possible degree of vision in growing eyes, because at Sydnexis, we believe that any diopter lost is one too many.
Leading the Charge in Pediatric Ophthalmology
We are a mission-driven team of scientists, clinicians, and innovators dedicated to transforming pediatric eye care.
Founded in 2014 and based in San Diego, Sydnexis is privately held and backed by Visionary Ventures, RA Capital, Longitude Capital, and Bluestem Capital. We are proud to be advancing science that has the potential to protect children’s sight.

We are guided by a team of experts working to transform the future of children’s eye care.
Join the Mission to Protect Children’s Sight
"*" indicates required fields
